Sugar coating has long been the traditional method of coating pharmaceutical dosage forms. The process has its origin in the confectionery industry and has been used in the pharmaceutical industry since the late 19th century. The process of sugar coating involves the successive deposition of aqueous sugar solution on the tablet cores as they are rotated and tumbled in a revolving pan by spraying sugar solution or suspensions into pans and drying off the solvent.
Sugar coating is used in immediate release applications to mask unpleasant taste and odour of some drugs or to improve aesthetic qualities of the product. It should be understood that the coating process will add some time to the overall disintegration of the tablet and may impact drug dissolution. This effect should be considered when formulating the core to ensure that the product meets disintegration and dissolution requirements stated in official compendia. For an enteric or sustained-release sugar-coated product, the formulation problem may become more complex to meet USP tablet disintegration and dissolution specifications. The selection of the core tablet and coating materials becomes more important for these applications and requires proper evaluation to assure long-term chemical and physical stability.
Sugar coatings are essentially aqueous based, unless a seal coat (e.g., alcoholic confectioners glaze) is required to protect the core tablet from water used in the sugar-coating process. Although the sugar-coating process is lengthy and labour intensive, coating materials are inexpensive and readily available.
Contents
Because sugar-coating process consists of various steps, a variety of additives may be incorporated into each type of formulation to achieve a particular function. These include:
As expected, the major ingredient used in sugar-coating process is sugar (primarily sucrose), although this may be substituted by other sugars and sugar alcohols (such as glucose, lactose, maltitol, mannitol, isomalt, sorbitol, xylitol, and sugar mixtures such as invert sugar and starch sugars) for low calorie diabetic products (typically in the candy industry) and for the fact that sucrose cause dental caries.
Sucrose is used in concentrations ranging between 50-60%, since syrups with a sugar content of less than 65% are stable at room temperature without crystallization occurring.
Binders increase the strength and elasticity of the coating by forming bonds and thus a coherent matrix. Examples of binders used in sugar coating include polyvinyl acetate (PVA), polyvinyl pyrrolidone (PVP), carboxymethyl starch, dextrin, acacia gum, gelatin, agar-agar, sodium alginate, cellulose ethers, and starches.
Fillers build up the structure and adds mass to the coatings. E.g., precipitated calcium carbonate, talc, kaolin, dextrin, powdered acacia, corn starch, and calcium sulfate.
Colourants add color to the coatings and cover imperfections which may appear in the tablet core. Examples include dyes, lakes (aluminum lakes), and pigments (titanium dioxide or other inorganic coloring agents).
These materials reduce friction between coated tablets and thus prevent dust formation during drying. Examples include talc and colloidal silicon dioxide.
Flavors improve and enhance the acceptability and palatability of the dosage form in order to maximize patient compliance. E.g., cinnamon, fruit flavors, etc.
They prevent phase separation or sedimentation of the coating suspension while it is being applied during the coating process. Examples include surface-active agents (emulsifying agents, bentonite) or thickening agents.
These materials smoothen out the surface of the coatings by lubricating and binding the fines that may be created during the coating process. Example includes a combination of syrup and acacia gum.
Polishing agents enhance the reflectivity and intensity of the colour of the coatings. Beeswax and carnauba wax are good examples of polishing agents used in sugar coating process.
The three techniques commonly used in sugar-coating process include:
This technique involves the application of syrup at room temperature. This coating technique includes 3 steps:
Note that the time required for distribution and drying is critical to obtain a smooth even coating.
Compared to the plain sugar-coating technique, the lamination coating is a more complicated technique involving two steps:
In order to obtain a reasonable weight increase within a short period of time, adjustment must be made between powder and liquid quantities and process performed by skillful operators.
As suggested by the name, hot sugar coating simply involves the application of heated syrup on tablet core. The syrup is heated above room temperature to reduce the viscosity of the syrup. Attempts to prevent crystallization of sugar during processing may make this technique more complicated and more expensive since all equipment parts used must be insulated and heated.
Sugar-coating process consists of various steps, each designed to achieve a particular function. A typical sugar-coating process encompasses six stages:
Read more on steps involved in sugar-coating of tablets
Read more on sugar-coating defects, causes, and remedies
Owing to the amount of material applied as a coating in the sugar-coating process, it is not appropriate to grind up reject sugar-coated tablets for recompaction. One potentially viable recovery procedure (although one not without its difficulties because of handling problems) is to wash off the sugar coating by carefully dipping the coated tablets (held on a screen) into a water bath until sufficient coating is removed such that on subsequent refinishing, the desired quality is achievable. Once the requisite quantity of coating is removed, the tablets can be dried by tumbling in a coating pan under a warm air stream (50°C). Such a procedure must obviously be validated to ensure that overall product quality is not compromised.
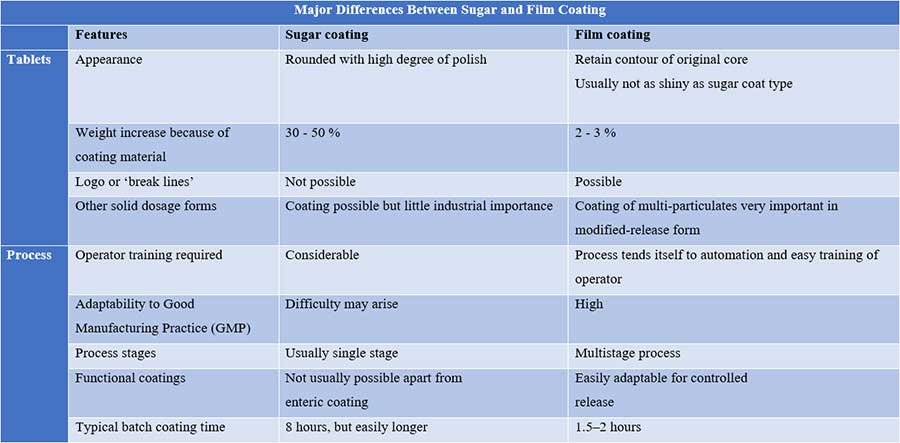
While the popularity of sugar coating has certainly declined due to the advent of film-coated tablets (as a result of the improved mechanical properties of the latter coating), this process is still used by many companies that have invested in the complete modernization of the process.
Related keywords: sugar coating examples, sugar coating synonym, literary term for sugar coating, sugar coating meme, word for sugar coating the truth, is sugar coating lying, sugar-coated tongue meaning,sugar coating process slideshare